 
Given the electric field, the strength and direction of a force F on a quantity charge q in an electric field E is determined by the electric field. As the magnitude of opposing charges increases, energy increases and ionic bonding is more favorable.Īn electric field is a vector field which associates to each point of the space the Coulomb force that will experience a test unity charge. Generally, as the distance between ions increases, the energy of attraction approaches zero and ionic bonding is less favorable. This simple law also correctly accounts for the forces that bind atoms together to form molecules and for the forces that bind atoms and molecules together to form solids and liquids. No exceptions have ever been found, even at the small distances within the atom.Ĭoulomb’s law holds even within the atoms, correctly describing the force between the positively charged nucleus and each of the negatively charged electrons. For example, it has been shown that the force is inversely proportional to distance between two objects squared (F∝1/r 2) to an accuracy of 1 part in 1016. Modern experiments have verified Coulomb’s law to great precision. 
It is more easily affected by electrostatic forces than molecules with uniform charge distributions. The electrons spend more time near the oxygen than the hydrogens, giving a permanent charge separation as shown. The mathematical formula for the electrostatic force is called Coulomb ‘s law after the French physicist Charles Coulomb (1736–1806), who performed experiments and first proposed a formula to calculate it.Ĭharge distribution in a water molecule: Schematic representation of the outer electron cloud of a neutral water molecule. The Superposition Theorem finds use in the study of alternating current (AC) circuits, and semiconductor (amplifier) circuits, where sometimes AC is often mixed (superimposed) with DC. Through the work of scientists in the late 18th century, the main features of the electrostatic force -the existence of two types of charge, the observation that like charges repel, unlike charges attract, and the decrease of force with distance-were eventually refined, and expressed as a mathematical formula. 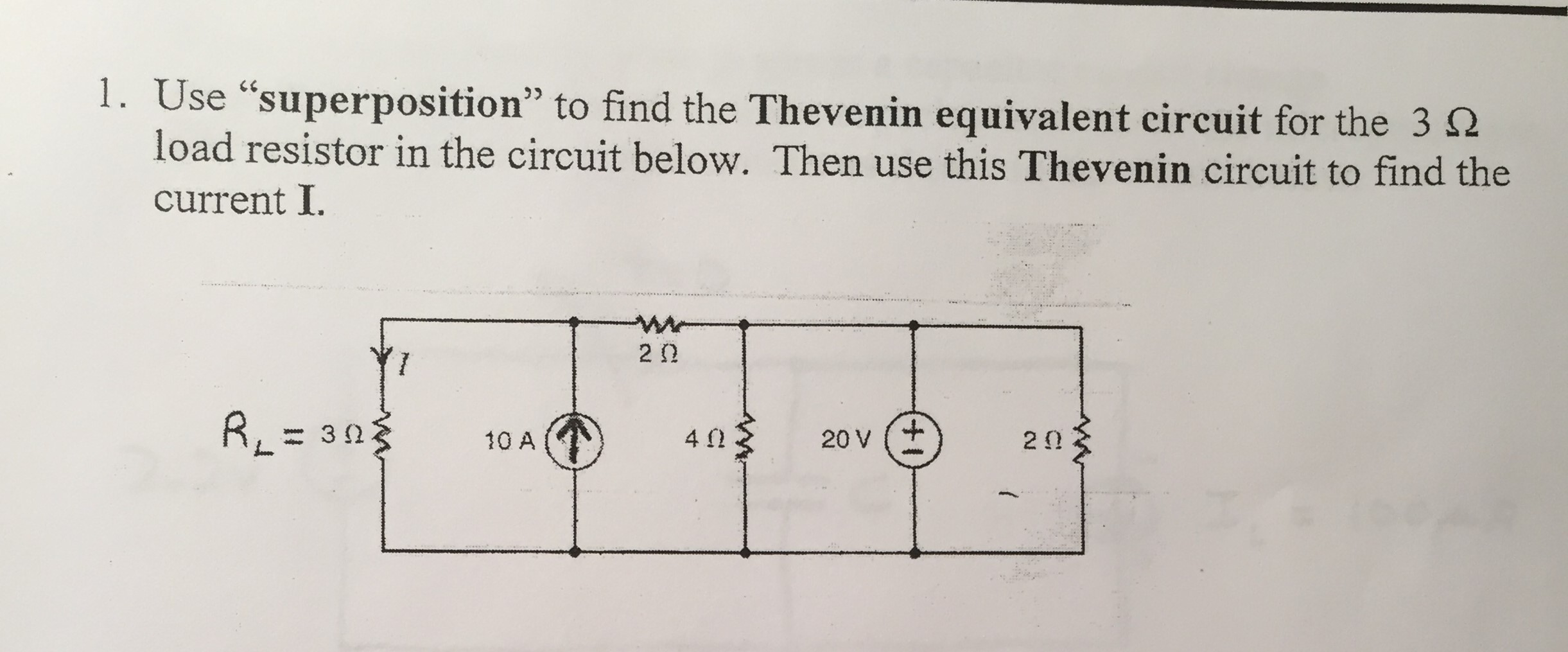
Describe shape of a Coulomb force from a spherical distribution of charge.(In this particular example of the moving charge, the force due to the presence of electromagnetic field is collectively called Lorentz force (see ). Total force, affecting the motion of the charge, will be the vector sum of the two forces. 
For example, when a charge is moving in the presence of a magnetic field as well as an electric field, the charge will feel both electrostatic and magnetic forces. Of course, our discussion of superposition of forces applies to any types (or combinations) of forces. \) (a vector pointing from charges q i to q. Ts = A0.sv 44 Vo Te Nw = O-S a x Tk = Ty -2 on ate eo ay TS -4%e-1tI 7 FO aio a eam ee = ears ei 7 Te A, 13☂5 So eee eS OPS ee ae WS PX $s T2 I (31 ae Sa Yee Br ee = Lop 1 pt,-ela tv =O Cop 2 Ie = 24 I2 Tg ~ 12T% e VFO Lee 3 1x D5 ~6T4-Gy =O Lop 3924 -GT,- PT2 =O eT y + 2) ~ Aye “IV +w- Meo - fo T'4 fol’s = 220" q = 12 (oO be Ta So erry eta. © yA 2 a x oh Py Verily | Z-47T"4 ve0 AL leg 4 Leda 2- 4M r20"20 tep2% gh - 114 -V=0 aay? ec? (2993 Vie qt co eve 4p - (ep 2ly ~« Ty = ple LA mt ae = J zis o-Sh = S ai z Me kt a et Ves Viet V"x : nek B wW,= qT’. Download Circuit Analyis Using Superposition Theorem and more Exercises Electrical Circuit Analysis in PDF only on Docsity! Gerd AP" -Vso4 Ve2T” lep2 r-4p +V =O loop > 2 Bx - 24 =O aD. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
